NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Madame Curie Bioscience Database [Internet]. Austin (TX): Landes Bioscience; 2000-2013.
Introduction
The erbB2 (also known as HER2 or neu) gene encodes a 185-kDa transmembrane glycoprotein, which belongs to the epidermal growth factor receptor (EGFR) family. ErbB2 is a receptor tyrosine kinase with intrinsic tyrosine kinase activity. The mammalian EGFR family comprises four receptors (EGFR, ErbB2, ErbB3, and ErbB4), which are derived from a series of gene duplications early in vertebrate evolution and are 40%—45% identical.1 ErbB2 is the only EGFR family member for which no ligand has been found. This may be explained by the unique structure of the ErbB2 extracellular domain, which is not favorable for ligand binding.2,3 Since ErbB2 extracellular domain is always in the open conformation, ErbB2 is the preferred binding partner of all ErbB receptors even as a monomer.2-4 The binding of ErbB2 to other ErbB receptors results in increased signaling potency of the dimerized receptors through several means, including increased ligand affinity, increased coupling efficiency to signaling molecules, and decreased rate of receptor internalization.5-8
ErbB2 plays an important role in human malignancies. The erbB2 gene is amplified or overexpressed in approximately 30% of human breast cancers9 and in many other cancer types, including ovarian,9 stomach,10 bladder,11 salivary,12 and lung carcinomas.13 Overwhelming evidence from numerous studies indicates that amplification or overexpression of ErbB2 disrupts normal cell-control mechanisms and gives rise of aggressive tumor cells.14 Patients with ErbB2-overexpressing breast cancer have substantially lower overall survival rates and shorter disease-free intervals than patients whose cancer does not overexpress ErbB2. Moreover, overexpression of ErbB2 leads to increased breast cancer metastasis.15-17 The important roles of ErbB2 in cancer progression render it a highly attractive target for therapeutic interventions of breast cancer.18,19 The humanized ErbB2-targeting antibody trastuzumab (Herceptin, from Genentech)20 was approved for the treatment of ErbB2-overexpressing breast cancers in 1998. In both phase II and phase III clinical trials, the antibody has shown remarkable therapeutic efficacy when given in combination with chemotherapeutic agents.21,22 Trastuzumab represents an excellent example of the ErbB2-targeting therapies. Other efforts to develop ErbB2-targeting cancer therapies also have yielded promising results, such as E1A gene therapy,23,24 single-chain antibodies,25,26 and tyrosine kinase inhibitors,27 just to name a few.
These novel targeted cancer therapies provide exciting new hope and opportunity for fighting breast cancers. However, the majority of patients with breast carcinoma still receive chemotherapy as a critical component of multimodality treatment.28 Thus, understanding the effect and mechanisms of ErbB2 on chemosensitivity is important for anticancer agent selection and individualization of patient treatment, which are critical to the success of treatment of breast cancers.
ErbB2 and Chemoresistance
Although the role and the mechanisms of ErbB2 overexpression on chemosensitivity still require intensive investigation, findings of many clinical and laboratory studies suggest that ErbB2 overexpression leads to increased chemoresistance to certain chemotherapeutic agents.
Clinical Studies
In a clinical study of breast cancer patients receiving adjuvant chemotherapy (cyclophosphamide, methotrexate, 5-fluorouracil, and prednisone [CMFP]), those with ErbB2-negative tumors showed a significantly greater rate of disease free survival in response to therapy than patients with ErbB2-positive tumors, indicating that overexpression of ErbB2 may play a role in resistance to chemotherapy.29 In another study, it was found that tumors that overexpress ErbB2 are less responsive to cyclophosphamide, methotrexate, and 5-fluorouracil (CMF) adjuvant therapy regimen than tumors that express a normal amount of ErbB2 protein.30 ErbB2 overexpression was also shown to have predictive value in epirubicin therapy in patients with advanced breast cancer.31 It has been reported that tumors coexpressing ErbB2 and Ras proteins were less responsive to tamoxifen and CMF regimens than those expressing low levels of ErbB2 and Ras.32 Recently, several groups reported that an elevated serum ErbB2 level predicted an unfavorable response to hormonal therapy or chemotherapy in patients with advanced metastatic breast cancer.33-36 Supporting data also came from the clinical trials in which trastuzumab was combined with chemotherapeutic agents. In a phase III first-line study, the combination of trastuzumab and chemotherapy produced significantly greater clinical benefit than chemotherapy alone.37 Similarly, a randomized phase II study comparing trastuzumab plus docetaxel with docetaxel alone in 188 patients with metastatic breast cancer yielded a statistically significant difference in terms of overall response rate, time to treatment progression and overall survival in favor of the combination.38 These results suggest that ErbB2 overexpression plays a role in inducing chemoresistance.
Laboratory Findings
Data from laboratory studies have provided more direct evidence that ErbB2-overexpressing breast cancer cells are more resistant to certain chemotherapeutic agents than cells that do not overexpress ErbB2. A panel of human breast cancer cell lines expressing ErbB2 at different levels was tested for their sensitivity to paclitaxel and docetaxel.39 Higher expression of ErbB2 in these cell lines correlated well with resistance to the drugs, and downregulation of cell-surface ErbB2 using an anti-ErbB2 monoclonal antibody significantly sensitized the cell lines to paclitaxel. The results indicate that overexpression of ErbB2 renders human breast cancer cells resistant to paclitaxel.39 In another study, MDA-MB-435 human breast cancer cells40 were stably transfected with the erbB2 gene that led to increased ErbB2 expression and no change in the expression of the multidrug resistance gene mdr1, but the ErbB2-overexpressing transfectants were more resistant to paclitaxel and docetaxel than their parental cells.41 This leads to the conclusion that overexpression of ErbB2 can lead to intrinsic paclitaxel and docetaxel resistance independent of the mdr1-mediated multidrug resistance mechanism. These findings are supported by results of other independent studies using multiple breast cancer cell lines that express high levels of ErbB2 protein.42-46 Moreover, downregulation of ErbB2 with the antisense oligonucleotides in ErbB2-overexpressing BT-474 breast cancer cells suppressed ErbB2 overexpression by 60.5% and subsequently increased the sensitivity of these cells to adriamycin and paclitaxel 20.8- and 10.8-fold, respectively.47 Furthermore, several other ErbB2-targeting approaches that downregulate ErbB2, such as humanized anti-ErbB2 antibodies,48,49 tyrosine kinase inhibitors,50 and adenovirus type 5 E1A,51,52 all led to sensitization to certain chemotherapeutic agents in cultured cancer cells or in animal models. Taken together, these laboratory findings clearly indicate that ErbB2 overexpression is linked to resistance to particular chemotherapeutic agents.
The Existing Controversy
Despite the supporting evidence just described, current clinical and experimental data on the effect of ErbB2 overexpression on chemosensitivity remain controversial. Results of several clinical studies have suggested that ErbB2 overexpression does not necessarily lead to chemoresistance. One clinical study showed that ErbB2 expression does not predict response to docetaxel or sequential methotrexate and 5-fluorouracil in advanced breast cancers.53 Another study showed that ErbB2 expression was not significantly associated with tumor response to neoadjuvant treatment with fluorouracil, adriamycin and cyclophoshamide (FAC) in 329 cases of breast cancer.54 In yet another study, ErbB2-overexpressing breast cancers responded better to doxorubicin than did breast cancers expressing low levels of ErbB2.55
These contradictory clinical observations may be partly explained by intrinsic differences in the design of the studies. Since clinical studies are complicated processes, numerous factors may affect the outcome of the investigations. For example, amplification and overexpression of ErbB2 can be detected by fluorescence in situ hybridization, immunohistochemistry, or enzyme-linked immunosorbent assay on tumor tissue samples.56 However, use of nonstandardized assay, subjectivity of the assay performer, limitations of techniques, differences in antibodies or DNA probes used, and differences in tissue treatment procedure have resulted in discordance in determining ErbB2 expression levels. In other words, the way in which the ErbB2 expression level was determined and defined is a very important factor that may affect the outcome of a study. Other factors that may affect outcome include the timing of treatment (neoadjuvant or adjuvant); the type of regimen (e.g., FAC or CMF); the treatment status of the patient (previously treated or untreated); patient's age (younger or older), menopausal status, and ethnic background; estrogen-receptor status of the tumor (positive or negative); and presence or absence of other genetic alterations that may interact with the ErbB2 receptor. Thus, any of these factors may lead to discordance in the outcomes of investigations on the role of ErbB2 in modulating chemosensitivity.57
The controversy over the role of ErbB2 in chemosensitivity also exists in laboratory studies. For example, In a laboratory study in which erbB2-transfected breast and ovarian cancer cells were used to determine the effect of ErbB2 overexpression on chemosensitivity, ErbB2 overexpression was found to be insufficient to induce intrinsic resistance to drugs, including paclitaxel.58 Although this study involved the use of a series of erbB2-transfected breast cancer cells, including erbB2-transfected MDA-MB-435 breast cancer cells, it yielded results that apparently disagreed with those of an earlier study showing that ErbB2 can confer paclitaxel resistance in erbB2-transfected MDA-MB-435 cells.41 The discrepancy may be explained partly by differences in ErbB2 expression levels between the erbB2-transfected MDA-MB-435 cells used in the two studies. In particular, the erbB2 transfectants that showed paclitaxel resistance expressed very high levels of ErbB2 protein, similar to those expressed in the SKBR3 breast cancer cell line, which was established from an ErbB2-overexpressing primary breast tumor.41 However, the erbB2 transfectants that showed no paclitaxel resistance produced less than one-third of the level of ErbB2 protein expressed by SKBR3 cells.58,57 Based on the data from these two independent studies, we suggest that ErbB2 overexpression must reach a threshold level in breast cancer cells before they become resistant to paclitaxel. This might provide a reasonable explanation for the discrepancy between the findings of the two studies.
Molecular Mechanisms of ErbB2-Mediated Chemoresistance
The role of ErbB2 in chemoresistance is a problem of great clinical importance, and the observational data are abundant, as described above. Because of the controversy and the complexity of this problem, however, our knowledge on the molecular mechanisms of how ErbB2 confers chemoresistance is still limited. It is generally believed that breast cancer cells overexpressing ErbB2 are intrinsically resistant to DNA-damaging agents such as cisplatin as the result of an altered cell-cycle checkpoint, altered DNA repair mechanisms, and altered apoptosis responses.59,60
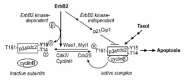
Apoptosis is a predominant mechanism by which cancer chemotherapeutic agents kill cells.61,62 The failure of cancer cells to detect chemotherapeutic agent-induced damage and to activate apoptosis may lead to multidrug resistance. The results from our studies indicate that overexpression of ErbB2 renders human breast cancer cells resistant to paclitaxel.39,41 While studying the molecular mechanisms of ErbB2-mediated paclitaxel resistance, we found that treatment of MDA-MB-435 breast cancer cells with paclitaxel caused them to undergo apoptosis, which was inhibited in their paired transfectants overexpressing ErbB2. Further investigation showed that paclitaxel-treatment induced a premature activation of p34Cdc2 kinase, the mitotic serine/threonine kinase that binds to cyclin B and also plays an important role in cancer cell apoptosis.63 The premature activation of p34Cdc2 led to mitotic catastrophe, i.e., apoptosis. A chemical inhibitor of p34Cdc2 and a dominant-negative mutant of p34Cdc2 blocked paclitaxel-induced apoptosis in these cells, indicating that a premature Cdc2 activation is a prerequisite for paclitaxel-induced apoptosis in MDA-MB-435 cells. We demonstrated that overexpression of ErbB2 in MDA-MB-435 cells transcriptionally upregulates p21Cip1, which associates with p34Cdc2, inhibits paclitaxel-mediated p34Cdc2 activation, delays cell entrance to G2/M phase, and thereby inhibits paclitaxel-induced apoptosis. Therefore, upregulation of p21Cip1 by ErbB2 inhibits p34Cdc2 and deregulates G2/M checkpoint that contributes to resistance to paclitaxel-induced apoptosis in ErbB2-overexpressing breast cancer cells.64 In addition, we found that phosphorylation on tyrosine (Tyr)15 of Cdc2, the crucial inhibitory phosphorylation site of Cdc2, is elevated in ErbB2-overexpressing breast cancer cells and primary tumors independent of Wee1, Cdc25C, and p21Cip1. We showed that ErbB2 binds to and colocalizes with cyclin B-Cdc2 complexes and can directly and specifically phosphorylate Cdc2 on Tyr15. Increased Cdc2-Tyr15 phosphorylation in ErbB2-overexpressing cells corresponds with delayed M phase entry and reduced sensitivity to paclitaxel-induced apoptosis. Expression of a kinase-defective ErbB2 in MDA-MB-435 breast cancer cells or expression of a nonphosphorylatable Cdc2Y15F mutant in ErbB2 gene transfectant of the MDA-MB-435 cells render the cells sensitive to paclitaxel-induced apoptosis. These data indicate that the increased Cdc2-Tyr15 phosphorylation by ErbB2 tyrosine kinase may be a pertinent cell-cycle checkpoint defect that is involved in paclitaxel resistance in ErbB2-overexpressing breast cancers. Taken together, these findings indicate that ErbB2 overexpression can confer breast cancer cell resistance to paclitaxel-induced apoptosis by inhibiting Cdc2 activation through at least two mechanisms: (1) ErbB2 kinase-independent p21 upregulation and (2) ErbB2 kinase-dependent direct phosphorylation of Cdc2 on Tyr15 (fig. 1). This model is further supported by results of a study on patient samples showing that ErbB2 overexpression is correlated with p21Cip1 upregulation and with increased p34Cdc2-Tyr15 phosphorylation in breast tumors.65
Other molecular mechanisms may also underlie ErbB2-mediated paclitaxel resistance. In addition, the molecular mechanisms of ErbB2-mediated resistance to different chemotherapeutic agents could be different. For example, other molecular mechanisms of ErbB2-mediated chemoresistance may involve activation of the PI3K/Akt pathway by ErbB2, which leads to increased cancer cell survival,46 the estrogen receptor-ErbB2 cross-talk in ErbB2-positive breast cancer cells,66,67 and coexpression of ErbB2 with other ErbB family receptors.44 Although hints of how ErbB2-overexpressing breast tumors evade chemotherapeutic agent-induced apoptosis and develop chemoresistance are beginning to surface, it is obvious that many questions remain to be answered.
Targeting ErbB2 to Overcome Chemoresistance
Numerous lines of laboratory and clinical evidence indicate that ErbB2-targeting agents can sensitize the response of tumor cells to chemotherapeutic agents; therefore, developing ErbB2-targeting strategies to improve the therapy of ErbB2-overexpressing breast cancer remains a high priority. During the last decade, several exciting techniques have been developed to target ErbB2. Although some are still under investigation, many studies have shown that these ErbB2-targeting techniques not only inhibit tumor growth, but also lead to chemosensitization of ErbB2-overexpressing cancer cells (Table 1).
Table 1
Possible ways to overcome ErbB2-mediated chemoresistance.
One of the most successful example is Trastuzumab, which is a humanized antibody that binds to the extracellular domain of ErbB2.20 Recent studies have shown that, in addition to inhibition of ErbB2 signaling, trastuzumab has other functions, such as activation of the PTEN tumor suppressor gene,68 induction of p27Kip1, and induction of G1 cell cycle arrest.69,70 Trastuzumab has demonstrated tumor-inhibitory and chemosensitizing effects for paclitaxel and several other chemotherapeutic agents in preclinical studies and in phase II and phase III clinical trials.21,22,48,71 These results represent an excellent example of anti-ErbB2 antibody-mediated chemosensitization. On the basis of our understanding of the mechanisms of ErbB2-mediated paclitaxel resistance, we investigated the mechanisms by which trastuzumab enhances the antitumor effects of paclitaxel in vitro and in vivo. We found that treatment of ErbB2-overexpressing cells with trastuzumab can inhibit ErbB2-mediated Cdc2-Tyr15 phosphorylation and p21Cip1 upregulation, which allows effective p34Cdc2 activation and induction of apoptosis upon paclitaxel treatment.49,65
In addition, the past 15 years have witnessed the development of several effective ErbB2-tageting strategies. These include, but not limited to, adenovirus type 5 E1A protein (please refer to the chapter by Liao and Hung for more details),51,72-75,76 ErbB2-specific tyrosine kinase inhibitors such as emodin,50 HKI-272,27 and GW572016,77 anti-ErbB2 intracellular single-chain antibodies (sFv),25,26,78,79 ErbB2-targeting antisense oligonucleotide,47,80-83 rationally designed anti-ErbB2 peptide mimetics (AHNP),84-86 all-trans retinoic acid (ATRA) and fenretinide (4-HPR).87 These ErbB2-tageting strategies either have been shown to have chemosensitization effect or are currently under testing for chemosensitization in ErbB2-overexpressing breast cancer cell lines, animal models, or clinical trials.57
These approaches discussed above have already shown promise in overcoming ErbB2-mediated chemoresistance in either laboratory or clinical studies. Other new technologies are also under development. For example, ErbB2-targeting small interfering RNAs (siRNAs) silence erbB2 mRNA by using double-stranded RNA oligonucleotides.88-90 One study showed that ovarian cancer cells and breast cancer cells infected with a retrovirus expressing anti-ErbB2 siRNA exhibited effective downregulation of ErbB2 expression and slower proliferation, increased apoptosis, increased G0/G1 arrest, and decreased tumor growth in mouse models.89 On the basis of these results, ErbB2-targeting siRNA may have great potential in overcoming ErbB2-mediated chemoresistance in ErbB2-overexpressing breast cancer cells. Additionally, attempts are being made to modulate existing chemotherapeutic agents so that they can overcome resistance.91 These combined efforts held great potential to improve the therapies for patients with ErbB2-overexpressing breast tumors.
Future Investigation
Understanding the role of ErbB2 in chemoresistance is important and has significant clinical relevance. Although many studies have been done during the last decade, our current knowledge on the role of ErbB2 in cancer chemosensitivity is still limited. To develop more effective treatment for patients with ErbB2-overexpressing breast cancers, scientists and physicians need to team up to pursue the following and other related issues.
Well Designed and Performed Laboratory and Clinical Studies
Current data from our studies and those of others indicate that ErbB2 renders breast cancers resistant to DNA-damaging agents such as cisplatin60 and to the microtubule-stabilizing agents taxenes.39,41 To better understand the role of ErbB2 in chemosensitivity, we must determine whether ErbB2 overexpression also renders breast cancers resistant to other chemotherapeutic agents frequently used for treating such cancers. This should be investigated (1) in carefully designed laboratory studies that use ErbB2-overexpressing breast cancer cell lines that express ErbB2 at levels similar to those detected in primary breast tumors and compare between cell lines having similar genetic backgrounds, and (2) in well-controlled and -defined large-scale clinical trials that can adequately assess the impact of the various factors important for the chemoresponse of tumors.
Other Possible Mechanisms for ErbB2-Mediated Chemoresistance
Many mechanisms of drug resistance in various tumor cells are well established, including enhanced drug metabolism, reduced drug accumulation, drug target amplification, and repair of damaged targets and apoptosis resistance.61 Our data indicate that in breast cancer cells, ErbB2 can block paclitaxel-induced apoptosis by upregulating p21cip1 and by directly phosphorylating the inhibitory tyrosine on p34Cdc2. Both mechanisms lead to the inhibition of p34Cdc2 kinase, which is required for paclitaxel-induced apoptosis. It has also been reported that chemotherapeutic agents-induced apoptosis depends on a balance between cell-cycle checkpoints and DNA-repair mechanisms. In addition, downstream signaling of the ErbB2 receptor tyrosine kinase, e.g., Akt activation, also plays a critical role in ErbB2-mediated chemoresistance.46 However, the downstream signal-transduction cascades by which ErbB2 affects chemosensitivity still require more investigation. Most ErbB2-overexpressing breast cancers also express other ErbB receptors, which can form heterodimers with ErbB2. An interconnected network of ErbB signaling pathways may be activated by the overexpression of ErbB2, and that determines tumor cell response to chemotherapeutic stress. It is, therefore, important to perform in-depth investigations into the role of ErbB2 receptor signaling in the regulation of stress-responsive genes. A better understanding of other possible mechanisms underlying ErbB2-mediated chemoresistance is also critical to the development of better alternative therapeutic strategies to overcome intrinsic chemoresistance.
Tailoring Therapeutic Strategies to Individual Patients
One of the important aspects of future research efforts on ErbB2-mediated chemoresistance is to identify other unknown genetic alterations that interact with ErbB2 and contribute to ErbB2-mediated chemoresistance. ErbB2-mediated chemoresistance is a very complicated problem, as it may be specific to tumor, cell, chemotherapeutic agent, regimen, timing, population, or patient age.57 With today's powerful genomic, proteomic, kinomic, and tissue microarray technologies, it has become feasible to identify factors that interact with ErbB2 and contribute to ErbB2-mediated chemoresistance from tumors of individual patient. This information can then be used to develop therapeutic strategies that are tailored for individual patient and are based on the ErbB2 biology and any identified relevant factors. These individually tailored therapeutic strategies will greatly maximize the therapeutic benefit for individual breast cancer patients.
New Approaches for Overcoming ErbB2-Mediated Chemoresistance
We have discussed several promising strategies for overcoming ErbB2-mediate chemoresistance (Table 1). New and more effective strategies are still in demand and can be developed. Recently, nanotechnology has emerged as a promising strategy for drug delivery. Nanotechnology-based delivery systems can be used to achieve cellular or tissue targeted drug delivery, to improve drug bioavailability, to sustain drug effect in target tissue, to solubilize drugs for intravascular delivery, and to improve the stability of therapeutic agents.92 With nanotechnology-based delivery systems, chemotherapeutic agents such as paclitaxel, doxorubicin, and 5-fluorouracil, have been shown to achieve improved efficacy to inhibit tumor growth in vitro and in vivo.93,94 In addition, nanotechnology-based system has been shown to potentially improve the antitumor efficacy of ErbB2-targeting agents, such as trastuzumab.95 It is foreseeable that by using these new techniques, which will produce highly efficient chemotherapeutic agents and ErbB2-targeting strategies, we may conquer ErbB2-mediated chemoresistance more effectively.
Acknowledgements
We apologize to those whose work could not be cited owing to space limitations. We thank Katherine Hale for editorial assistance. D. Yu's laboratory is supported by NIH grants RO1-CA60488, RO1-CA109570, 1PO1-CA099031-1 project 4, and the Department of Defense grant DAMD17-02-1-0462.
References
- 1.
- Stein RA, Staros JV. Evolutionary analysis of the ErbB receptor and ligand families. J Mol Evol. 2000;50(5):397–412. [PubMed: 10824084]
- 2.
- Cho HS, Mason K, Ramyar KX. et al. Structure of the extracellular region of HER2 alone and in complex with the Herceptin Fab. Nature. 2003;421(6924):756–760. [PubMed: 12610629]
- 3.
- Garrett TP, McKern NM, Lou M. et al. The crystal structure of a truncated ErbB2 ectodomain reveals an active conformation, poised to interact with other ErbB receptors. Mol Cell. 2003;11(2):495–505. [PubMed: 12620236]
- 4.
- Graus-Porta D, Beerli RR, Daly JM. et al. ErbB-2, the preferred heterodimerization partner of all ErbB receptors, is a mediator of lateral signaling. EMBO J. 1997;16:1647–1655. [PMC free article: PMC1169769] [PubMed: 9130710]
- 5.
- Pinkas-Kramarski R, Soussan L, Waterman H. et al. Diversification of Neu differentiation factor and epidermal growth factor signaling by combinatorial receptor interactions. EMBO J. 1996;15:2452–2467. [PMC free article: PMC450177] [PubMed: 8665853]
- 6.
- Riese IInd DJ, van RaaijTM, Plowman GD. et al. The cellular response to neuregulins is governed by complex interactions of the erbB receptor family. Mol Cell Biol. 1995;15(10):5770–5776. [PMC free article: PMC230829] [PubMed: 7565730]
- 7.
- Karunagaran D, Tzahar E, Beerli RR. et al. ErbB-2 is a common auxiliary subunit of NDF and EGF receptors: Implications for breast cancer. EMBO J. 1996;15:254–264. [PMC free article: PMC449940] [PubMed: 8617201]
- 8.
- Jones JT, Akita RW, Sliwkowski MX. Binding specificities and affinities of egf domains for ErbB receptors. FEBS Lett. 1999;447(2-3):227–231. [PubMed: 10214951]
- 9.
- Slamon DJ, Godolphin W, Jones LA. et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989;244(4905):707–712. [PubMed: 2470152]
- 10.
- Lemoine NR, Jain S, Silvestre F. et al. Amplification and overexpression of the EGF receptor and c-erbB-2 proto-oncogenes in human stomach cancer. Br J Cancer. 1991;64(1):79–83. [PMC free article: PMC1977330] [PubMed: 1677259]
- 11.
- Sauter G, Moch H, Moore D. et al. Heterogeneity of erbB-2 gene amplification in bladder cancer. Cancer Res. 1993;53(10 Suppl):2199–2203. [PubMed: 8097962]
- 12.
- Stenman G, Sandros J, Nordkvist A. et al. Expression of the ERBB2 protein in benign and malignant salivary gland tumors. Genes Chromosomes Cancer. 1991;3(2):128–135. [PubMed: 1676907]
- 13.
- Tateishi M, Ishida T, Mitsudomi T. et al. Prognostic value of c-erbB-2 protein expression in human lung adenocarcinoma and squamous cell carcinoma. Eur J Cancer. 1991;27(11):1372–1375. [PubMed: 1683776]
- 14.
- Hung M-C, Schechter AL, Chevray P-YM. et al. Molecular cloning of the neu gene: absence of gross structural alteration in oncogenic alleles. Proc Natl Acad Sci USA. 1986;83:261–264. [PMC free article: PMC322837] [PubMed: 3001730]
- 15.
- Tan M, Yao J, Yu D. Overexpression of the c-erbB-2 gene enhanced intrinsic metastatic potential in human breast cancer cells without increasing their transformation abilities. Cancer Res. 1997;57:1199–1205. [PubMed: 9067293]
- 16.
- Moody SE, Sarkisian CJ, Hahn KT. et al. Conditional activation of Neu in the mammary epithelium of transgenic mice results in reversible pulmonary metastasis. Cancer Cell. 2002;2(6):451–461. [PubMed: 12498714]
- 17.
- Holbro T, Civenni G, Hynes NE. The ErbB receptors and their role in cancer progression. Exp Cell Res. 2003;284(1):99–110. [PubMed: 12648469]
- 18.
- Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol. 2001;2(2):127–137. [PubMed: 11252954]
- 19.
- Shawver LK, Slamon D, Ullrich A. Smart drugs: Tyrosine kinase inhibitors in cancer therapy. Cancer Cell. 2002;1(2):117–123. [PubMed: 12086869]
- 20.
- Carter P, Presta L, Gorman CM. et al. Humanization of an anti-p185HER2 antibody for human cancer therapy. Proc Natl Acad Sci USA. 1992;89(10):4285–4289. [PMC free article: PMC49066] [PubMed: 1350088]
- 21.
- Pegram MD, Lipton A, Hayes DF. et al. Phase II study of receptor-enhanced chemosensitivity using recombinant humanized anti-p185HER2/neu monoclonal antibody plus cisplatin in patients with HER2/neu-overexpressing metastatic breast cancer refractory to chemotherapy treatment. J Clin Oncol. 1998;16(8):2659–2671. [PubMed: 9704716]
- 22.
- Dickman S. Antibodies stage a comeback in cancer treatment. Science. 1998;280:1196–1197. [PubMed: 9634400]
- 23.
- Hortobagyi GN, Ueno NT, Xia W. et al. Cationic liposome-mediated E1A gene transfer to human breast and ovarian cancer cells and its biologic effects: A phase I clinical trial. J Clin Oncol. 2001;19(14):3422–3433. [PubMed: 11454891]
- 24.
- Madhusudan S, Tamir A, Bates N. et al. A multicenter Phase I gene therapy clinical trial involving intraperitoneal administration of E1A-lipid complex in patients with recurrent epithelial ovarian cancer overexpressing HER-2/neu oncogene. Clin Cancer Res. 2004;10(9):2986–2996. [PubMed: 15131034]
- 25.
- Alvarez RD, Barnes MN, Gomez-Navarro J. et al. A cancer gene therapy approach utilizing an anti-erbB-2 single-chain antibody-encoding adenovirus (AD21): A phase I trial. Clin Cancer Res. 2000;6(8):3081–3087. [PubMed: 10955787]
- 26.
- Azemar M, Djahansouzi S, Jager E. et al. Regression of cutaneous tumor lesions in patients intratumorally injected with a recombinant single-chain antibody-toxin targeted to ErbB2/HER2. Breast Cancer Res Treat. 2003;82(3):155–164. [PubMed: 14703062]
- 27.
- Rabindran SK, Discafani CM, Rosfjord EC. et al. Antitumor activity of HKI-272, an orally active, irreversible inhibitor of the HER-2 tyrosine kinase. Cancer Res. 2004;64(11):3958–3965. [PubMed: 15173008]
- 28.
- Buchholz TA, Hunt KK, Whitman GJ. et al. Neoadjuvant chemotherapy for breast carcinoma: Multidisciplinary considerations of benefits and risks. Cancer. 2003;98(6):1150–1160. [PubMed: 12973838]
- 29.
- Allred DC, Clark GM, Tandon AK. et al. Her-2/neu in node-negative breast cancer: prognostic significance of overexpression influenced by the presence of in situ carcinoma. J Clin Oncol. 1992;10:599–605. [PubMed: 1548522]
- 30.
- Gusterson BA, Gelber RD, Goldhirsch A. et al. Prognostic importance of c-erbB-2 expression in breast cancer. J Clin Oncol. 1992;10(7):1049–1056. [PubMed: 1351538]
- 31.
- Jarvinen TA, Holli K, Kuukasjarvi T. et al. Predictive value of topoisomerase II alpha and other prognostic factors for epirubicin chemotherapy in advanced breast cancer. Br J Cancer. 1998;77:2267–2273. [PMC free article: PMC2150425] [PubMed: 9649144]
- 32.
- Giani C, Finocchiaro G. Mutation rate of the CDKN2 gene in malignant gliomas. Cancer Res. 1994;54(24):6338–6339. [PubMed: 7987825]
- 33.
- Fehm T, Maimonis P, Katalinic A. et al. The prognostic significance of c-erbB-2 serum protein in metastatic breast cancer. Oncology. 1998;55:33–38. [PubMed: 9428373]
- 34.
- Colomer R, Montero S, Lluch A. et al. Circulating HER2 extracellular domain and resistance to chemotherapy in advanced breast cancer. Clin Cancer Res. 2000;6:2356–2362. [PubMed: 10873087]
- 35.
- Classen S, Kopp R, Possinger K. et al. Clinical relevance of soluble c-erbB-2 for patients with metastatic breast cancer predicting the response to second-line hormone or chemotherapy. Tumour Biol. 2002;23(2):70–75. [PubMed: 12065844]
- 36.
- Colomer R, Llombart-Cussac A, Lluch A. et al. Biweekly paclitaxel plus gemcitabine in advanced breast cancer: Phase II trial and predictive value of HER2 extracellular domain. Ann Oncol. 2004;15(2):201–206. [PubMed: 14760109]
- 37.
- Slamon DJ, Leyland-Jones B, Shak S. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344(11):783–792. [PubMed: 11248153]
- 38.
- Extra JM, Cognetti F, Chan S. et al. First-line trastuzumab (Herceptin') plus docetaxel versus docetaxel alone in women with HER2-positive metastatic breast cancer (MBC): Results from a randomised phase II trial (M77001). Br Cancer Res Treat. 2003;82(suppl 1):S47.
- 39.
- Yu D, Liu B, Jing T. et al. Overexpression of both p185c-erbB2 and p170mdr-1 renders breast cancer cells highly resistant to taxol. Oncogene. 1998;16(16):2087–2094. [PubMed: 9572489]
- 40.
- Sellappan S, Grijalva R, Zhou X. et al. Lineage infidelity of MDA-MB-435 cells: Expression of melanocyte proteins in a breast cancer cell line. Cancer Res. 2004;64(10):3479–3485. [PubMed: 15150101]
- 41.
- Yu D, Liu B, Tan M. et al. Overexpression of c-erbB-2/neu in breast cancer cells confers increased resistance to Taxol via mdr-1-independent mechanisms. Oncogene. 1996;13:1359–1365. [PubMed: 8808711]
- 42.
- Sabbatini AR, Basolo F, Valentini P. et al. Induction of multidrug resistance (MDR) by transfection of MCF-10A cell line with c-Ha-ras and c-erbB-2 oncogenes. Int J Cancer. 1994;59(2):208–211. [PubMed: 7927921]
- 43.
- Ciardiello F, Caputo R, Pomatico G. et al. Resistance to taxanes is induced by c-erbB-2 overexpression in human MCF-10A mammary epithelial cells and is blocked by combined treatment with an antisense oligonucleotide targeting type I protein kinase A. Int J Cancer. 2000;85(5):710–715. [PubMed: 10699953]
- 44.
- Chen X, Yeung TK, Wang Z. Enhanced drug resistance in cells coexpressing ErbB2 with EGF receptor or ErbB3. Biochem Biophys Res Commun. 2000;277(3):757–763. [PubMed: 11062025]
- 45.
- Witters LM, Santala SM, Engle L. et al. Decreased response to paclitaxel versus docetaxel in HER-2/ neu transfected human breast cancer cells. Am J Clin Oncol. 2003;26(1):50–54. [PubMed: 12576925]
- 46.
- Knuefermann C, Lu Y, Liu B. et al. HER2/PI-3K/Akt activation leads to a multidrug resistance in human breast adenocarcinoma cells. Oncogene. 2003;22(21):3205–3212. [PubMed: 12761490]
- 47.
- Tanabe K, Kim R, Inoue H. et al. Antisense Bcl-2 and HER-2 oligonucleotide treatment of breast cancer cells enhances their sensitivity to anticancer drugs. Int J Oncol. 2003;22(4):875–881. [PubMed: 12632082]
- 48.
- Baselga J, Norton L, Albanell J. et al. Recombinant humanized anti-HER2 antibody (Herceptin®) enhances the antitumor activity of paclitaxel and doxorubicin against HER2/neu overexpressing human breast cancer xenografts. Cancer Res. 1998;58:2825–2831. [PubMed: 9661897]
- 49.
- Lee S, Yang W, Lan KH. et al. Enhanced sensitization to taxol-induced apoptosis by herceptin pretreatment in ErbB2-overexpressing breast cancer cells. Cancer Res. 2002;62(20):5703–5710. [PubMed: 12384528]
- 50.
- Zhang L, Lau YK, Xia W. et al. Tyrosine kinase inhibitor emodin suppresses growth of HER-2/ neu-overexpressing breast cancer cells in athymic mice and sensitizes these cells to the inhibitory effect of paclitaxel. Clin Cancer Res. 1999;5:343–353. [PubMed: 10037184]
- 51.
- Ueno NT, Yu D, Hung MC. Chemosensitization of HER-2/neu-overexpressing human breast cancer cells to paclitaxol (Taxol) by adenovirus type 5 E1A. Oncogene. 1997;15:953–960. [PubMed: 9285690]
- 52.
- Ueno NT, Bartholomeusz C, Xia W. et al. Systemic gene therapy in human xenograft tumor models by liposomal delivery of the E1A gene. Cancer Res. 2002;62(22):6712–6716. [PubMed: 12438271]
- 53.
- Sjostrom J, Collan J, von Boguslawski K. et al. C-erbB-2 expression does not predict response to docetaxel or sequential methotrexate and 5-fluorouracil in advanced breast cancer. Eur J Cancer. 2002;38(4):535–542. [PubMed: 11872346]
- 54.
- Rozan S, Vincent-Salomon A, Zafrani B. et al. No significant predictive value of c-erbB-2 or p53 expression regarding sensitivity to primary chemotherapy or radiotherapy in breast cancer. Int J Cancer. 1998;79:27–33. [PubMed: 9495354]
- 55.
- Porter-Jordan K, Lippman ME. Overview of the biologic markers of breast cancer. Breast Cancer. 1994;8:73–100. [PubMed: 8150784]
- 56.
- Ross JS, Fletcher JA, Bloom KJ. et al. HER-2/neu testing in breast cancer. Am J Clin Pathol. 2003;120(Suppl):S53–71. [PubMed: 15298144]
- 57.
- Yu D, Hung M-C. Role of erbB2 in breast cancer chemosensitivity. BioEssays. 2000;22:673–680. [PubMed: 10878580]
- 58.
- Pegram MD, Finn RS, Arzoo K. et al. The effect of HER2/neu overexpression on chemotherapeutic drug sensitivity in human breast and ovarian cancer cells. Oncogene. 1997;15:537–547. [PubMed: 9247307]
- 59.
- Pietras RJ, Pegram MD, Finn RS. et al. Remission of human breast cancer xenografts on therapy with humanized monoclonal antibody to HER-2 receptor and DNA-reactive drugs. Oncogene. 1998;17:2235–2249. [PubMed: 9811454]
- 60.
- Alaoui-Jamali MA, Paterson J, Al Moustafa AE. et al. The role of erbB-2 tyrosine kinase receptor in cellular intrinsic chemoresistance: Mechanisms and implications. Biochem Cell Biol. 1997;75:315–325. [PubMed: 9493954]
- 61.
- Fisher DE. Apoptosis in cancer therapy: Crossing the threshold. Cell. 1994;78:539–542. [PubMed: 8069905]
- 62.
- Wang CY, Cusack JrJC, Liu R. et al. Control of inducible chemoresistance: Enhanced anti-tumor therapy through increased apoptosis by inhibition of NF-kappaB. Nat Med. 1999;5(4):412–417. [PubMed: 10202930]
- 63.
- Gao CY, Zelenka PS. Induction of cyclin B and H1 kinase activity in apoptotic PC12 cells. Exp Cell Res. 1995;219:612–618. [PubMed: 7641812]
- 64.
- Yu D, Jing T, Liu B. et al. Overexpression of ErbB2 blocks Taxol-induced apoptosis by upregulation of p21Cip1, which inhibits p34Cdc2 kinase. Mol Cell. 1998;2(5):581–591. [PubMed: 9844631]
- 65.
- Yang W, Klos KS, Zhou X. et al. ErbB2 overexpression in human breast carcinoma is correlated with p21Cip1 up-regulation and tyrosine-15 hyperphosphorylation of p34Cdc2: Poor responsiveness to chemotherapy with cyclophoshamide methotrexate, and 5-fluorouracil is associated with Erb2 overexpression and with p21Cip1 overexpression. Cancer. 2003;98(6):1123–1130. [PubMed: 12973835]
- 66.
- Osborne CK, Bardou V, Hopp TA. et al. Role of the estrogen receptor coactivator AIB1 (SRC-3) and HER-2/neu in tamoxifen resistance in breast cancer. J Natl Cancer Inst. 2003;95(5):353–361. [PubMed: 12618500]
- 67.
- Shou J, Massarweh S, Osborne CK. et al. Mechanisms of tamoxifen resistance: Increased estrogen receptor-HER2/neu cross-talk in ER/HER2-positive breast cancer. J Natl Cancer Inst. 2004;96(12):926–935. [PubMed: 15199112]
- 68.
- Nagata Y, Lan KH, Zhou X. et al. PTEN activation contributes to tumor inhibition by trastuzumab, and loss of PTEN predicts trastuzumab resistance in patients. Cancer Cell. 2004;6(2):117–127. [PubMed: 15324695]
- 69.
- Le XF, Claret FX, Lammayot A. et al. The role of cyclin-dependent kinase inhibitor p27Kip1 in anti-HER2 antibody-induced G1 cell cycle arrest and tumor growth inhibition. J Biol Chem. 2003;278(26):23441–23450. [PubMed: 12700233]
- 70.
- Nahta R, Takahashi T, Ueno NT. et al. P27(kip1) down-regulation is associated with trastuzumab resistance in breast cancer cells. Cancer Res. 2004;64(11):3981–3986. [PubMed: 15173011]
- 71.
- Baselga J, Tripathy D, Mendelsohn J. et al. Phase II study of weekly intravenous recombinant humanized anti-p185HER2 monoclonal antibody in patients with HER2/neu-overexpressing metastatic breast cancer. J Clin Oncol. 1996;14(3):737–744. [PubMed: 8622019]
- 72.
- Yu D, Scorsone K, Hung M-C. Adenovirus type 5 E1A gene products act as transformation suppressors of the neu oncogene. Mol Cell Biol. 1991;11(3):1745–1750. [PMC free article: PMC369488] [PubMed: 1671710]
- 73.
- Yu DH, Hamada JI, Zhang H. et al. Mechanisms of c-erbB2/neu oncogene-induced metastasis and repression of metastatic properties by adenovirus 5 E1A gene products. Oncogene. 1992;7:2263–2270. [PubMed: 1359495]
- 74.
- Yu D, Suen T-C, Yan D-H. et al. Transcriptional repression of the neu protooncogene by the adenovirus 5 E1A gene products. Proc Natl Acad Sci USA. 1990;87(12):4499–4503. [PMC free article: PMC54143] [PubMed: 1972274]
- 75.
- Yu D, Matin A, Xia W. et al. Liposome-mediated in vivo E1A gene transfer suppressed dissemination of ovarian cancer cells that overexpress HER-2/neu. Oncogene. 1995;11:1383–1388. [PubMed: 7478560]
- 76.
- Liao Y, Zou YY, Xia WY. et al. Enhanced paclitaxel cytotoxicity and prolonged animal survival rate by a nonviral-mediated systemic delivery of E1A gene in orthotopic xenograft human breast cancer. Cancer Gene Ther. 2004;11(9):594–602. [PubMed: 15272313]
- 77.
- Spector NL, Xia W, Burris IIIrd H. et al. Study of the biological effects of lapatinib, a reversible inhibitor of ErbB1 and ErbB2 tyrosine kinases, on tumor growth and survival pathways in patients with advanced malignancies. J Clin Oncol. 2005 (Epub ahead of print) [PubMed: 15684311]
- 78.
- Deshane J, Loechel F, Conry RM. et al. Intracellular single-chain antibody directed against erbB2 down-regulates cell surface erbB2 and exhibits a selective anti-proliferative effect in erbB2 overexpressing cancer cell lines. Gene Ther. 1994;1(5):332–337. [PubMed: 7584099]
- 79.
- Barnes MN, Deshane JS, Siegal GP. et al. Novel gene therapy strategy to accomplish growth factor modulation induces enhanced tumor cell chemosensitivity. Clin Cancer Res. 1996;2:1089–1095. [PubMed: 9816272]
- 80.
- Rait AS, Pirollo KF, Xiang L. et al. Tumor-targeting, systemically delivered antisense HER-2 chemosensitizes human breast cancer xenografts irrespective of HER-2 levels. Mol Med. 2002;8(8):475–486. [PMC free article: PMC2040013] [PubMed: 12435858]
- 81.
- Rait AS, Pirollo KF, Ulick D. et al. HER-2-targeted antisense oligonucleotide results in sensitization of head and neck cancer cells to chemotherapeutic agents. Ann NY Acad Sci. 2003;1002:78–89. [PubMed: 14751825]
- 82.
- Waterhouse DN, Dragowska WH, Gelmon KA. et al. Pharmacodynamic behavior of liposomal antisense oligonucleotides targeting Her-2/neu and vascular endothelial growth factor in an ascitic MDA435/LCC6 human breast cancer model. Cancer Biol Ther. 2004;3(2):197–204. [PubMed: 14726684]
- 83.
- Funato T, Kozawa K, Fujimaki S. et al. Increased sensitivity to cisplatin in gastric cancer by antisense inhibition of the her-2/neu (c-erbB-2) gene. Chemotherapy. 2001;47(4):297–303. [PubMed: 11399867]
- 84.
- Park BW, Berezov A, Wu CW. et al. Rationally designed anti-HER2/neu peptide mimetic disables p185HER2/neu tyrosine kinases in vitro and in vivo. Nat Biotech. 2000;18:194–198. [PubMed: 10657127]
- 85.
- Berezov A, Chen J, Liu Q. et al. Disabling receptor ensembles with rationally designed interface peptidomimetics. J Biol Chem. 2002;277(31):28330–28339. [PubMed: 12011054]
- 86.
- Murali R, Liu Q, Cheng X. et al. Antibody like peptidomimetics as large scale immunodetection probes. Cell Mol Biol. 2003;49(2):209–216. [PubMed: 12887103]
- 87.
- Grunt T, Dittrich E, Ofterdinger M. et al. Effects of retinoic acid and fenretinide on the c-erbB-2 expression, growth and cisplatin sensitivity of breast cancer cells. Br J Cancer. 1998;78:79–87. [PMC free article: PMC2062943] [PubMed: 9662255]
- 88.
- Mellinghoff IK, Vivanco I, Kwon A. et al. HER2/neu kinase-dependent modulation of androgen receptor function through effects on DNA binding and stability. Cancer Cell. 2004;6(5):517–527. [PubMed: 15542435]
- 89.
- Yang G, Cai KQ, Thompson-Lanza JA. et al. Inhibition of breast and ovarian tumor growth through multiple signaling pathways by using retrovirus-mediated small interfering RNA against Her-2/neu gene expression. J Biol Chem. 2004;279(6):4339–4345. [PubMed: 14625284]
- 90.
- Choudhury A, Charo J, Parapuram SK. et al. Small interfering RNA (siRNA) inhibits the expression of the Her2/neu gene, upregulates HLA class I and induces apoptosis of Her2/neu positive tumor cell lines. Int J Cancer. 2004;108(1):71–77. [PubMed: 14618618]
- 91.
- Hayes DF, Henderson IC, Shapiro CL. Treatment of metastatic breast cancer: Present and future prospects. Seminars in Oncol. 1995;22(2):5–21. [PubMed: 7537913]
- 92.
- Panyam J, Labhasetwar V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv Drug Deliv Rev. 2003;55(3):329–347. [PubMed: 12628320]
- 93.
- BrannonPeppas L, Blanchette JO. Nanoparticle and targeted systems for cancer therapy. Adv Drug Deliv Rev. 2004;56(11):1649–1659. [PubMed: 15350294]
- 94.
- Garber K. Improved Paclitaxel formulation hints at new chemotherapy approach. J Natl Cancer Inst. 2004;96(2):90–91. [PubMed: 14734692]
- 95.
- Ito A, Kuga Y, Honda H. et al. Magnetite nanoparticle-loaded anti-HER2 immunoliposomes for combination of antibody therapy with hyperthermia. Cancer Lett. 2004;212(2):167–175. [PubMed: 15279897]
- Molecular Mechanisms of ErbB2-Mediated Breast Cancer Chemoresistance - Madame Cu...Molecular Mechanisms of ErbB2-Mediated Breast Cancer Chemoresistance - Madame Curie Bioscience Database
- The Role of Neuropilin in Vascular and Tumor Biology - Madame Curie Bioscience D...The Role of Neuropilin in Vascular and Tumor Biology - Madame Curie Bioscience Database
- DNA Vaccines against Bacterial Pathogens - Madame Curie Bioscience DatabaseDNA Vaccines against Bacterial Pathogens - Madame Curie Bioscience Database
- Genetic Risk Factors for Disseminated Intravascular Coagulation - Madame Curie B...Genetic Risk Factors for Disseminated Intravascular Coagulation - Madame Curie Bioscience Database
- Regulation of Cardiomyocyte Proliferation and Apoptosis - Madame Curie Bioscienc...Regulation of Cardiomyocyte Proliferation and Apoptosis - Madame Curie Bioscience Database
Your browsing activity is empty.
Activity recording is turned off.
See more...