Included under terms of UK Non-commercial Government License.
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Duffy SW, Mackay J, Thomas S, et al. Evaluation of mammographic surveillance services in women aged 40–49 years with a moderate family history of breast cancer: a single-arm cohort study. Southampton (UK): NIHR Journals Library; 2013 Mar. (Health Technology Assessment, No. 17.11.)

Evaluation of mammographic surveillance services in women aged 40–49 years with a moderate family history of breast cancer: a single-arm cohort study.
Show detailsBasic design and end points
FH01 was designed as a single-arm cohort study (see Appendix 1 for protocol and Appendix 2 for protocol of the accompanying blood study). The intervention to be evaluated was annual mammography (or at any rate, with the interval not slipping beyond 18 months) for 5 years. We targeted women aged 40–44 years at recruitment so that after 5 years of mammography they would still be in the age range 40–49 years. This was to avoid arguments about ‘age creep’: the theory propounded about randomised trials of screening that the apparent benefits of screening in women aged 40–49 years at randomisation were actually due to screening activity taking place after the recruits had passed their 50th birthdays.20 It should be noted that there is little empirical evidence for the phenomenon,21,24 but it was considered prudent to head off the issue by design if possible.
It was specified that, to be eligible, women had to satisfy at least one of the following family history criteria:
- one first-degree female relative with breast cancer diagnosed at ≤ 40 years of age
- one first-degree female relative with bilateral breast cancer diagnosed at < 50 years of age
- two first-degree or one first- and one second-degree female relative, both with breast cancer diagnosed at ≤ 60 years of age (same side of family)
- one first- or second-degree female relative with breast and ovarian cancer, with the first cancer diagnosed at ≤ 60 years of age
- three first- or second-degree female relatives with breast or ovarian cancer at any age (same side of family)
- one first-degree male relative with breast cancer at any age
- paternal history of a minimum of two second-degree relatives (father's first-degree relatives) with breast cancer at ≤ 50 years of age, or one with breast cancer at ≤ 50 years of age and an ovarian cancer (any age), or paternal uncle/grandfather with breast cancer at < 50 years of age.
A first-degree female relative is defined as mother, sister or daughter. A second-degree female relative is defined as granddaughter, grandmother, aunt or niece. Exclusion criteria were:
- inability to give written informed consent
- pregnancy
- age < 40 years
- proven breast cancer or ductal carcinoma in situ (DCIS)
- previous bilateral prophylactic mastectomy
- presence of a breast cancer type 1 (BRCA1) or breast cancer type 2 (BRCA2) mutation in the family in women who have been tested negative for the mutation.
On the basis of the inclusion criteria, the study group was anticipated to have at least a 3% probability of breast cancer between ages 40 and 49 years, inclusive. Although BRCA1- and BRCA2-positive cases were not explicitly excluded, the moderate-risk criteria implied that relatively few FH01 recruits would be BRCA positive.
The information sheet for potential recruits is given as Appendix 3. The information for primary care professionals is given in Appendix 4.
For reasons noted in Chapter 1, the primary end points were the size, node status and histological grade of tumours diagnosed and the projected mortality from these. The primary comparison group was the control group of the Age Trial. We planned to adjust the comparison for differences between the FH01 cohort and the Age Trial population in underlying risk of breast cancer. The adjustment was made by calculating the expected 10-year absolute incidence of breast cancer17 for each population (FH01 and Age Trial controls), and dividing the projected rate of breast cancer for each group by its expected incidence. We also planned to compare the FH01 results with those from other, historical family history cohorts undergoing little or no surveillance.25 Further details are given in Planned statistical analyses.
Collaborating units were expected to offer annual (or at least 18-monthly) two-view mammography and to:
- operate a breast cancer unit in line with the recommendations of the British Breast Group and the BASO guidelines for surgeons in the treatment of symptomatic breast disease26
- have experience in mammography in symptomatic women aged < 50 years
- either participate in the NHSBSP or offer mammographic services at a level consistent with the quality standards set out by the NHSBSP
- have a clearly defined referral line for high-risk women to a regional clinical genetics service
- have at least one member of the multidisciplinary team trained in pedigree construction and interpretation, and risk analysis.
Study size
We had originally designed FH01 to have the power for the same comparison of tumour attributes and the consequent expected mortality in two risk-stratified subgroups. As a result, the planned sample size at the initiation of the study was 10,000. A monitoring visit by the funding body noted that recruitment had been poor in the early years, and recommended changing the target to a more modest study size designed to have adequate power for the cohort as a whole, with no regard for subgroup analyses. For this target, assuming use of the controls in the Age Trial as a comparison group, an important planned comparison was the incidence of node-positive tumours in the FH01 cohort with that expected from the comparison group, taking into account the different underlying incidences in the two groups. From the Swedish Two-County Study controls, we would expect an unscreened tumour series in the age group 40–49 years to be node-positive in 42% of cases.27 In the Age Trial control group, with 7 years of cancer incidence in 106,000 women,22 we conservatively expected around 742 cancers, and therefore 311 (42%) node-positive tumours.
Results from the Two-County Study suggest a screening sensitivity of 83% and a mean sojourn time (average duration of the preclinical screen-detectable period) of 2.44 years in women aged 40–49 years.24 This suggests that with a 1-year interval there would be 77% screen-detected cancers, of which 11% would be node positive. We assume that the interval cancers would have the same 42% node-positive cancer rate as an unscreened group, giving an overall 18% node-positive cancer rate. Thus, the comparison anticipated is between a group with 42% node-positive cancers and one with 18% node-positive cancers. This would correspond, on the basis of the relative fatality of node-positive and node-negative cancers, to long-term survival of 64% compared with 74%, with a relative risk (RR) in the FH01 cohort of 0.72. A 5-year incidence rate at around 4 per 1000 per year (due to high familial risk) would mean a total incidence of node-positive disease of 3.6 per 1000 (0.18 × 0.004 × 5). For 90% power to detect a difference in incidence of node-positive cancers of 3.6 per 1000 and 8.4 per 1000 (0.0036 × 0.42/0.18) as significant, and allowing a 5% increase in standard errors as a result of adjustment for different underlying risk in the two populations, we would require 6000 women and 120 cancers. Thus, we aimed to recruit 6000 women and expected to be in a position to analyse the data and report after an average of 5 years' observation.
Planned statistical analyses
The data proforma is given in Appendix 5. As noted above, the primary analyses planned were the comparison of the prognostic variables tumour size, lymph node status and histological grade, and the consequent predicted breast cancer mortality, between the FH01 cohort and the Age Trial controls. Secondary comparisons with other historical data sets were anticipated. Categorical variables were compared between the FH01 tumours and comparison groups using the chi-squared test. Continuous variables were compared using the t-test. We calculated the Nottingham Prognostic Index (NPI) score for invasive cases as a + b + c, where
- a = 0.2 × size in cm
- b = 1 if node-negative, 2 if 1–3 positive nodes, 3 if ≥ 4 positive nodes; and
- c = histological grade (1, 2 or 3).
From this, we estimated the 10-year survival as shown by Blamey et al.,28 who regressed 10-year survival on NPI, splitting their data set in two for cross-validation purposes. They obtained two quadratic equations for the prediction of survival from NPI, which gave very similar predictions. Here we use the average of their two equations, giving an estimate S of average 10-year per cent survival as a function of NPI, denoted N in equation (1).
For the FH01 cohort and the Age Trial control group, we then calculated the absolute expected rate of tumours proving fatal over 10 years, and divided this in each case by the underlying risk in the two populations, calculated from family history and other risk-factor data using the absolute risk model of Tyrer et al.17 The Tyrer et al.17 model has been independently validated and shown to predict risk with accuracy.29 The absolute risk was calculated directly on all of the FH01 recruits. For the Age Trial comparison group, for the ethics and governance reasons outlined in Chapter 1, a more indirect process had to be used. There were no risk-factor data on the Age Trial control group, and there were ethical problems with contacting members of this group to ascertain risk factors. However, in another, unrelated study, a subset of the Age Trial intervention group had undergone risk factor ascertainment. We therefore used this study population to estimate the underlying risk in the Age Trial control group, on the basis that, owing to the randomisation, the risk profiles of members of the study group would be the same as those of the control group.
Thus, we were able to calculate the RR of absolute mortality, corrected for the different risk profiles of the two populations. This was done by dividing the expected death rates by Tyrer et al.'s17 independent estimates of the underlying incidence. The corrected RR was:
where d1, P1 and r1 are the expected deaths, person-years of observation and underlying 10-year average risk in the FH01 cohort, and d2, P2 and r2 the corresponding quantities in the Age Trial control group. The numerator and denominator of the RR are simply the ratios of expected death rates from the NPI to the expected incidences based on Tyrer et al.'s17 model, to adjust for the fact that the FH01 cohort has higher underlying risk than the Age Trial population. Again, note that r1 was calculated directly from the FH01 subjects, whereas r2 was calculated from a subgroup of 3001 members of the study group of the Age Trial. Note that the division by the expected incidence figures, r1 and r2, calculated from an independently derived algorithm, is a safeguard against lead time, length bias and overdiagnosis. Actual incidence in the FH01 cohort would have been potentially susceptible to these biasing factors, but independently estimated incidence is not.
The variance of the logarithm of RR is:
where f is the predicted case fatality rate from the NPI, that is, the complement of the predicted survival, and n is the number of invasive cancer cases in FH01.
The standard error of the logarithm of RR was then calculated as s, the square root of the variance and the 95% confidence interval (CI) on ln(RR) calculated as:
The end points of the interval were then transformed exponentially to give the 95% CI on RR.
In addition to the Age Trial controls, we also compared the FH01 results with those from a Dutch series of 238 breast cancers (all invasive) in women largely not subject to surveillance, with a family history of breast cancer and not BRCA1 or BRCA2 positive.25 However, for the Dutch series, we had only published tabular results. We did not have risk-factor data on which to calculate the absolute risks and correct the comparison for these.
From the published tabular data on tumour size, node status and grade in the Dutch study, we calculated average NPI and, to be conservative, the maximum possible standard error on this consistent with the tabular data. The predicted average 10-year survival and its standard error in the Dutch study were calculated from the average NPI, using the second-order Taylor approximation.30
It had originally been planned to compare our results with those of a French series of tumours in women with a family history who had not been subject to mammographic surveillance. However, preliminary results suggested that geographical and temporal confounding factors were rendering the comparison unreliable, in that our results would have seemed too good to be true. For example, 62% of the tumours in the French series were of size > 20 mm. We therefore abandoned this planned analysis.
One further analysis of clinical outcome was performed. From the rates of screen-detected and interval cancers by node status, we estimated the parameters of progression by node status in a Markov process. From these we estimated the cancers by node status which would have been expected to occur if the surveillance had not taken place. These were then combined with 20-year fatality rates of cancer by node status in the Swedish Two-County Trial of breast screening27 to estimate long-term mortality from breast cancer with and without mammographic surveillance. These results were used in the health economic analysis.
Other analyses planned included summaries of screening activity and negative outcomes of the surveillance, including false-positives, benign surgery cases and radiation dose. The study was accompanied by a psychosocial evaluation, which was funded separately and has already been published in detail.31–34 FH01 also incorporated radiology and pathology reviews, and an economic evaluation. These are also briefly reported on in the following chapters.
Recruitment
Recruitment took place between 16 January 2003 and 28 February 2007, with a total of 6710 women in the study. Figure 1 shows recruitment over time. A number of centres remained ineligible for the study because of an ongoing policy of 2-yearly surveillance. Recruitment began slowly, but accelerated steadily. In the first year, around 600 women were recruited, in the second year, around 1200, and in each of the third and fourth years > 2000. A number of measures were taken to improve recruitment during the course of the study, including expansion of the eligible centres to include Scotland and Northern Ireland, personal visits and other contacts to potentially high-recruitment centres and the institution of regional co-ordinators to take local responsibility for recruitment and data capture. Although these measures did bear fruit in terms of improved accrual, it is not clear which particular measures were the most effective. It is likely that if we had taken these measures from the study's inception, recruitment would have been considerably faster. However, the pattern of slow recruitment in the early months, gradually accelerating, is common in large population studies.
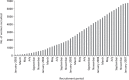
FIGURE 1
FH01 cumulative recruitment over time.
Table 1 shows recruitment by individual centre. Interestingly, Scotland, Wales and Northern Ireland contributed around 30% of recruits. Within England, major recruiting centres were the Withington Community Hospital Manchester, Nottingham City Hospital and the Jarvis Breast Screening and Diagnostic Centre, Guildford. In Wales, women were recruited in a single genetics service covering the entire country and within a single research network, although surveillance took place at three sites.
TABLE 1
Final recruitment figures from each screening centre
It should be noted that the slow recruitment in the first 2 years was not because of unwillingness of eligible women to participate in the study. In five centres polled, four reported that participation rates in excess of 90%, and one of 65%. The phenomenon derived from a combination of the delay in centres joining the study, largely because of ethics and governance formalities, and the fact that many centres had relatively few eligible women.
The comparison populations
The Age Trial randomised 53,890 (study group) women aged 40–42 in the general population years to invitation to annual mammography for 7 years, and 106,971 (control group) women of the same age to usual care.22 Recruitment took place between 1991 and 1999. As noted above, our aim was to compare the pathological characteristics and the corresponding predicted 10-year mortality between the cancers diagnosed in FH01 and those diagnosed in the control group of the Age Trial, during 622,127 person-years of follow-up. Thus, our comparison group would be of similar ages to the FH01 population and would not have been offered mammographic surveillance. The group would, however, have general population risk, whereas the FH01 population would be at enhanced risk of breast cancer owing to family history. To adjust for this, we used risk-factor data on 3001 subjects within the Age Trial study group, assuming equal underlying risk between the study and the control group due to the randomisation. We estimated the 10-year probabilities of breast cancer in the Age Trial and FH01 recruits, and corrected our estimate of relative mortality for these.
The Dutch comparison series comprised 238 breast cancers in women with a family history of the disease but without a known BRCA mutation, during the period 1980–2004. Ages at diagnosis ranged from 25 to 77 years. The cases were from a population largely not undergoing surveillance. Thus, the Dutch series was comparable with FH01 with respect to family history, and was mostly not subject to mammography. However, the age range was much wider in the Dutch series.
- Design, planned analysis and study size - Evaluation of mammographic surveillanc...Design, planned analysis and study size - Evaluation of mammographic surveillance services in women aged 40–49 years with a moderate family history of breast cancer: a single-arm cohort study
- Results of the systematic review - A systematic review and individual patient da...Results of the systematic review - A systematic review and individual patient data meta-analysis of prognostic factors for foot ulceration in people with diabetes: the international research collaboration for the prediction of diabetic foot ulcerations (PODUS)
- List of abbreviations - Plasma exchange and glucocorticoids to delay death or en...List of abbreviations - Plasma exchange and glucocorticoids to delay death or end-stage renal disease in anti-neutrophil cytoplasm antibody-associated vasculitis: PEXIVAS non-inferiority factorial RCT
- Conclusions - Home telemonitoring or structured telephone support programmes aft...Conclusions - Home telemonitoring or structured telephone support programmes after recent discharge in patients with heart failure: systematic review and economic evaluation
- Conclusions - Saline in Acute Bronchiolitis RCT and Economic evaluation: hyperto...Conclusions - Saline in Acute Bronchiolitis RCT and Economic evaluation: hypertonic saline in acute bronchiolitis – randomised controlled trial and systematic review
Your browsing activity is empty.
Activity recording is turned off.
See more...