Research evidence
It was not possible to use trial evidence to assess the effects of antenatal corticosteroids in women with growth-restricted fetuses, as the 2020 Cochrane review on antenatal corticosteroid efficacy did not conduct a subgroup analysis in this group. Any such subgroup analysis is unlikely to be informative as fetal growth restriction is often an exclusion criterion in many of these trials, but it is noteworthy that fetal growth restriction was not an exclusion criterion in the largest trial of this meta-analysis.
Evidence supporting the WHO 2015 recommendation was derived from a non-Cochrane systematic review of maternal and child outcomes following antenatal corticosteroid therapy of women in specific populations at risk of imminent preterm birth, which was subsequently published in 2016 (18). The review was updated in 2021 (19).
The updated review included a total of 18 cohort studies (8464 women and neonates) involving women with babies that were small for gestational age (SGA) and/or had fetal growth-restriction (FGR), who received antenatal corticosteroids compared to those who did not receive them. Additional unpublished data on one of the studies was obtained from a previous review paper identified through the search strategy.
Studies were conducted exclusively in high-income settings – three in the Netherlands, two each in Israel, Italy, the Republic of Korea and the United States of America; one each in Canada, China, France, Japan and Sweden; one study in Australia and New Zealand; and one study in Canada and the United States of America.
All women recruited into the studies were expected to give birth preterm (≤35 weeks) due to either spontaneous preterm labour, preterm prelabour rupture of the membranes or provider-initiated preterm birth due to fetal or maternal indications.
Three studies included babies with fetal growth restriction or who were small for gestational age (see Table 2), 12 studies included only small-for-gestational-age babies (see Table 3) and two included only babies with fetal growth restriction (see Table 4). One study reported fetal growth restriction and small-for-gestational-age separately. Where heterogeneity between the three sub-groups (i.e. SGA/FGR, SGA only, FGR only) was low, sub-group data were combined and total odds ratios calculated (see Table 1).
The studies all evaluated the use of a corticosteroid compared with no treatment. Eight studies used betamethasone in the treatment arm (1366 infants), one used dexamethasone (82 infants) and three studies used either betamethasone or dexamethasone (789 infants). The remaining six studies did not specify the type of corticosteroid used (6127 infants).
Sixteen studies specified that the comparison group comprised women who did not receive antenatal corticosteroids, one study compared antenatal corticosteroids to saline placebo and one compared to ‘no treatment’, without further explanation.
Antenatal corticosteroids versus placebo or no treatment — all growth-restricted babies (i.e. 18 studies: fetal growth restriction, small-for-gestational-age, and fetal growth restriction or small-for-gestational-age)
Download PDF (217K)
Maternal outcomes
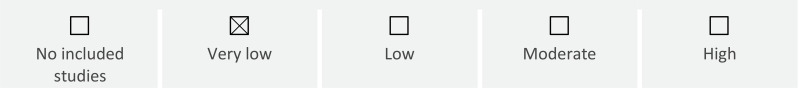
Maternal infectious morbidity: The evidence on the effect of antenatal corticosteroids on risk of chorioamnionitis among women with growth restricted babies is very uncertain No data were available for other maternal outcomes (maternal severe morbidity or death, adverse effects, well-being, satisfaction).
Infant outcomes
Fetal and neonatal death: The evidence on the effect of antenatal corticosteroids on risk of neonatal death before discharge among growth-restricted babies is very uncertain. No data were available on fetal death or perinatal death.
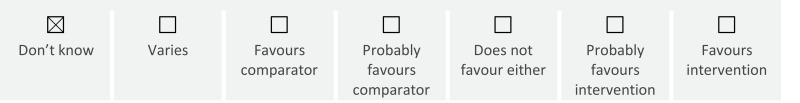
Severe neonatal morbidity: Among growth-restricted babies, antenatal corticosteroid therapy may increase the risk of neonatal hypoglycaemia
2 (OR 2.06 95% CI 1.27 to 3.32; 2 studies;
low certainty) and may reduce the risk of severe intraventricular haemorrhage (OR 0.54 95% CI 0.43 to 0.68; 9 studies;
low certainty) and duration of hospital stay (MD −2.32 95% CI −3.81 to −0.83; 2 studies;
low certainty).
The evidence on the effect of antenatal corticosteroid therapy on risk of intraventricular haemorrhage, neonatal infection, necrotizing enterocolitis, chronic lung disease/bronchopulmonary dysplasia, patent ductus arteriosus, retinopathy of prematurity and major brain lesion (IVH, Intracerebral haemorrhage [ICH] or PVL) among growth-restricted babies is very uncertain. No data were available on respiratory distress syndrome, moderate/severe respiratory distress syndrome, periventricular leukomalacia, surfactant use, admission to neonatal intensive care, use of mechanical ventilation, mean duration of mechanical ventilation.
Birth weight: No data were available for mean birth weight or low birth weight.
Long-term morbidity: The evidence on the effect of antenatal corticosteroid therapy and risk of cerebral palsy among growth-restricted babies is very uncertain. No data were available for other childhood or long-term outcomes (infant, child or adult death, developmental delay, intellectual, hearing or visual impairment, behavioural/learning difficulties).
Antenatal corticosteroids versus placebo or no treatment – babies with fetal growth restriction or small-for-gestational-age babies3 (i.e. three studies)
Download PDF (217K)
Maternal outcomes
Maternal infectious morbidity: The evidence on the effect of antenatal corticosteroids on risk of chorioamnionitis among women with FGR or SGA babies is very uncertain. No data were available on other maternal outcomes (severe maternal morbidity or death, adverse effects, maternal well-being, maternal satisfaction).
Infant outcomes
Fetal and neonatal death: The evidence on the effect of antenatal corticosteroids on risk of neonatal death among FGR or SGA babies is very uncertain. No data on perinatal death and fetal death were available.
Severe neonatal morbidity: Among FGR or SGA babies, antenatal corticosteroid therapy may reduce use of surfactant (OR 0.38 95% CI 0.23 to 0.62; 3 studies; low certainty), mechanical ventilation (OR 0.42 95% CI 0.26 to 0.66; 2 studies; low certainty) and use of oxygen therapy (OR 0.48 95% CI 0.30 to 0.77; 2 studies; low certainty) and duration of hospital stay (MD −2.32 95% CI −3.81 to −0.83; 1 study; low certainty).
The evidence on the effect of antenatal corticosteroids on risk of respiratory distress syndrome, intraventricular haemorrhage, severe intraventricular haemorrhage, neonatal infection, necrotizing enterocolitis, chronic lung disease/bronchopulmonary dysplasia, patent ductus arteriosus, retinopathy of prematurity, neonatal hypoglycaemia and admission to neonatal intensive care among FGR or SGA babies is very uncertain. No data were available for moderate/severe RDS or periventricular leukomalacia, admission to neonatal intensive care or mean duration of hospitalisation.
Birth weight: The evidence on the effect of antenatal corticosteroid therapy on mean birth weight among FGR or SGA babies is very uncertain. No data were available for low birth weight or small-for-gestational age.
Long-term morbidity: The evidence on the effect of antenatal corticosteroids therapy on survival free from disability and cerebral palsy among FGR or SGA babies is very uncertain. No data were available for other childhood or long-term outcomes (infant or child death, developmental delay, intellectual, hearing or visual impairment, behavioural/learning difficulties).
Antenatal corticosteroids versus placebo or no treatment – small-for-gestational-age babies only (i.e. 13 studies)
Download PDF (217K)
Maternal outcomes
Maternal infectious morbidity: The evidence on the effect of antenatal corticosteroid therapy on risk of chorioamnionitis among women with SGA babies is very uncertain.
No data were available on other maternal outcomes (severe maternal morbidity or death, adverse effects, maternal well-being, maternal satisfaction).
Infant outcomes
Fetal and neonatal death: Antenatal corticosteroid therapy may reduce the risk of neonatal death among SGA babies (OR 0.61 95% CI 0.49 to 0.78; 8 studies; low certainty). The evidence on the effect of antenatal corticosteroids on risk of death before discharge among SGA babies is very uncertain. No data were available on fetal death or perinatal death.
Severe neonatal morbidity: Antenatal corticosteroid therapy may reduce the risk of severe intraventricular haemorrhage among SGA babies (OR 0.52 95% CI 0.41 to 0.66; 7 studies; low certainty). The evidence on the effect of antenatal corticosteroid therapy on risk of respiratory distress syndrome, intraventricular haemorrhage, neonatal infection, necrotizing enterocolitis, chronic lung disease/bronchopulmonary dysplasia, patent ductus arteriosus, periventricular leukomalacia, retinopathy of prematurity, neonatal hypoglycaemia, major brain lesion (IVH, ICH or PVL), surfactant use, use of mechanical ventilation and duration of hospital stay among SGA babies is very uncertain. No data were available on moderate/severe respiratory distress syndrome, admission to neonatal intensive care or mean duration of mechanical ventilation.
Birth weight: The evidence on the effect of antenatal corticosteroids on mean birth weight among SGA babies is very uncertain. No data were available for low birth weight or small-for-gestational-age.
Long-term morbidity: The evidence on the effect of antenatal corticosteroids on risk of cerebral palsy, severe hearing impairment and visual impairment among SGA babies is very uncertain. No data were available on other childhood or long-term outcomes (infant or childhood death, developmental delay, intellectual impairment, behavioural/learning difficulties).
Antenatal corticosteroids versus placebo or no treatment – babies with fetal growth restriction only (i.e. three studies only)
Download PDF (278K)
Maternal outcomes
No data were available on maternal outcomes (severe maternal morbidity or death, maternal infectious morbidity, adverse effects, maternal well-being, maternal satisfaction).
Infant outcomes
Fetal and neonatal death: The evidence on the effect of antenatal corticosteroids on risk of neonatal death and risk of death before discharge among FGR babies is very uncertain. No data were available for fetal death or neonatal death.
Severe neonatal morbidity: The evidence on the effect of antenatal corticosteroids on risk of respiratory distress syndrome, intraventricular haemorrhage, severe intraventricular haemorrhage, neonatal sepsis, necrotizing enterocolitis, chronic lung disease/bronchopulmonary dysplasia, patent ductus arteriosus and major brain lesion (IVH, ICH or PVL), surfactant use and mean duration of mechanical ventilation among babies with fetal growth restriction is very uncertain. No data were available for moderate/severe respiratory distress syndrome, periventricular leukomalacia, retinopathy of prematurity, admission to neonatal intensive care, mean duration of hospitalization or use of mechanical ventilation.
Birth weight: The evidence on the effect of antenatal corticosteroids on small-for-gestational-age (<2.3rd percentile for gestational age) among FGR babies is very uncertain. No data were available for mean birth weight or low birth weight.
Long-term morbidity: The evidence on abnormal behaviour at long-term follow-up at school age among FGR babies is very uncertain. No data were available for other childhood or long-term outcomes (child death, cerebral palsy, developmental delay, intellectual, hearing or visual impairment, learning difficulties).
Additional considerations
Subgroup analyses involve splitting available trials into different groups of participants. However, it should be acknowledged that subgroup analyses are not based on randomized comparisons and are therefore susceptible to possible biases affecting observational studies (20).
The largest efficacy trial of antenatal corticosteroids in low-resource countries (WHO ACTION-I Trial) enrolled 2852 women, including 189 women with known or suspected fetal growth restriction (21).